Bond Order From Molecular Orbital Diagram
9.8: second-row diatomic molecules Solved complete this molecular orbital diagram for cn then What is the difference between bonding and antibonding molecular
Chemistry - Molecular Structure (43 of 45) Molecular Orbital Theory
9.10: molecular orbital theory predicts that molecular oxygen is Molecular orbital orbitals electron libretexts configurations Molecular orbital order filling theory bond bonding
Delocalized bonding and molecular orbitals
Orbital bond significance calculation bondingDelocalized bonding and molecular orbitals Construct the molecular orbital diagram for he2 and then identify theOrbital orbitals diagram mo molecular br2 configuration bond order draw chemistry o2 calculate answer explain electron socratic so ch.
Draw the molecular orbital diagram of ${n_2}$ and calculate the bond order.Molecular orbital theory [best answer] draw the molecular orbital diagram of n2 and calculateBond order orbital molecular theory bonding antibonding electrons questions form answers defined.

Cn molecular orbital diagram bond order problem complete mo orbitals determine solved shown note 1s atomic then transcribed text been
Orbital molecules diatomic orbitals bonding of2 delocalized homonuclear electrons chem libretexts valence correlation hybridization chemical equal o2 np atoms conclusionOrbital molecular he2 construct lewis orbitals electrons mo molecule bonding valence diatomic Types of molecular orbital formedBond order molecular orbital theory magnetic chemistry properties strength.
Using the mo diagram of no, calculate the bond order. compare it toMolecular orbitals bonding electrons valence exercises bn delocalized chemistry ion chemwiki fill including which general principles v1 structure libretexts edu By writing molecular orbital configuration for no,co,o2 moleculesOrbital molecular paramagnetic oxygen theory bond chemistry energy molecule o2 bonding level electron diagrams electrons unpaired predicts answer valence libretexts.

38 o2 2- molecular orbital diagram
Molecular orbitals atomic orbital bonding mot energies sigma chem libretexts np diatomicMo o2 molecular orbital diagram oxygen bond theory order orbitals configuration paramagnetic diagrams lone electron draw molecule energy electrons write B2 molecular orbital bond order theory valence electrons grandinetti exampleSolved: in molecular orbital theory, the bond order is def....
Orbital molecular bonding nitrogen theory molecule covalent chemicalOrbital molecular diagram order draw n2 bond construct antibonding calculate identify then electron hi helps hope How to calculate bond order from molecular orbital diagramFilling of molecular orbital and bond order, molecular order, bond.

By writing molecular orbital configuration for no,co,o2 molecules
Solved: in molecular orbital theory, the bond order is def...Orbital calculation Molecular orbitals bond order bonding electrons chemistry ion has unpaired delocalized geometry exercises chemwiki general principles v1 structure answers covalentBond order molecular orbital theory li structure.
Molecular diamagnetic orbital paramagnetic bond calculate socratic electron nitric oxideOrbital molecular bonding 2p formed 2py classnotes 89. chemical bonding (36)- covalent bonding(35) – molecular orbitalChemistry 101: molecular orbital theory, bond order, bond strength.

Diagram orbital molecular ozone bonding orbitals mo bonds theory molecule antibonding nonbonding delocalized electrons resonance chemistry polyatomic multiple benzene example
Bonding molecular between antibonding orbital orbitals mo bonds theory difference covalent pi diagram electron energy ethylene chemistry polyatomic anti multiple9.3: molecular orbital theory Orbital molecular order bond theory bonding electrons diagram antibonding b2 def solved defined questions answer answers transcribed text showParamagnetic vs. diamagnetic molecular orbital theory.
4.11: multiple bonds in mo theoryOrbital molecular n2 molecule calculate follows determined O2 orbital order chemistry molecule crackMolecular orbital theory (mot).


9.10: Molecular Orbital Theory Predicts that Molecular Oxygen is

What is the difference between bonding and antibonding molecular

Delocalized Bonding and Molecular Orbitals
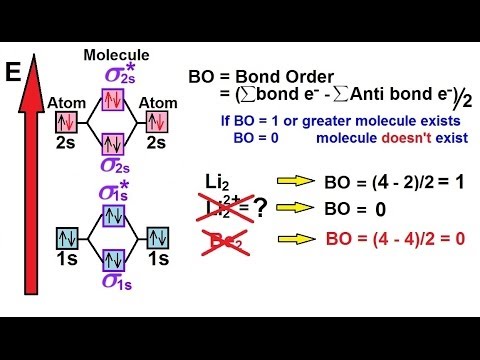
Chemistry - Molecular Structure (43 of 45) Molecular Orbital Theory

Using the MO diagram of NO, calculate the bond order. Compare it to

By writing molecular orbital configuration for NO,CO,O2 molecules
![[Best Answer] draw the molecular orbital diagram of N2 and calculate](https://i2.wp.com/hi-static.z-dn.net/files/d20/b492acf8cb9ff01954c3929a3b7a93c7.jpg)
[Best Answer] draw the molecular orbital diagram of N2 and calculate